Bringing
greater vision
to everyone
Individuelle OP-Sets in der Ophthalmologie
MED – Ihr Spezialist ist einer der weltweit führenden Hersteller von Abdecksystemen und OP-Sets aus Deutschland. Absolute Qualität, schnell, flexibel und zertifiziert, genau das Richtige für Ihre Operationsräume.
Qualität, 100% Made in Germany
- Eigene Entwicklung, immer „neu”
- Eigene Herstellung, auf neuestem Stand der Technik
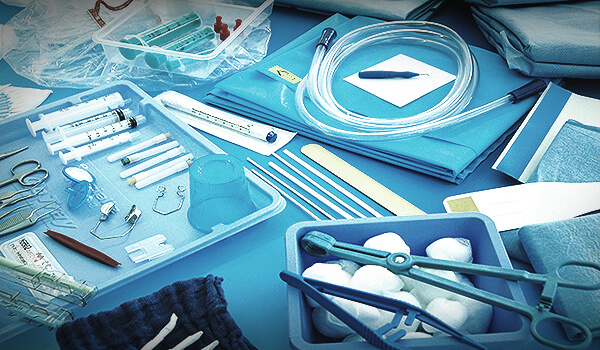
- Individuelle OP-Komplettsets: Sie stellen sie zusammen, wir stellen sie her
- Basis-OP-Komplettsets, bewährt seit knapp 20 Jahren
- Zertifiziertes Qualitätsmanagement
- Kürzeste Produktionszeit
- Top Lieferservice